Water oxidation—a critical yet sluggish step in green hydrogen production—is a major bottleneck for electrolysis efficiency. Traditional catalysts often degrade quickly under the high current densities needed for industrial scale. Researchers from the CAS Shanghai Institute of Ceramics (SIC), with collaborators, have unveiled a breakthrough catalyst designed for both high activity and remarkable durability. Published in Science on April 25, their innovative design starts as a MOF@POM superstructure, crafted by grafting cobalt-iron metal-organic frameworks (MOF) onto nickel-bridged polyoxometalates (POMs). During the water oxidation process, the MOF component transforms in-situ into a stable, single-layer cobalt-iron layered double hydroxide covalently bonded to the POM units. In-situ analysis revealed that its exceptional stability stems from a dual mechanism—dynamic electron tuning by the POM units and strain relief via covalent nickel-oxygen bonds. This catalyst requires only 178 mV overpotential at 10 mA/cm2 and powers an electrolyzer achieving 3 A/cm2 at 1.78 V—surpassing the U.S. Department of Energy’s 2025 industrial target. Crucially, it operates stably for 5,140 hours at room temperature with negligible decay and endures over 2,000 hours at 60°C. This design framework bridges lab-scale innovation to industrial-scale water splitting, propelling high-current, low-energy hydrogen production.
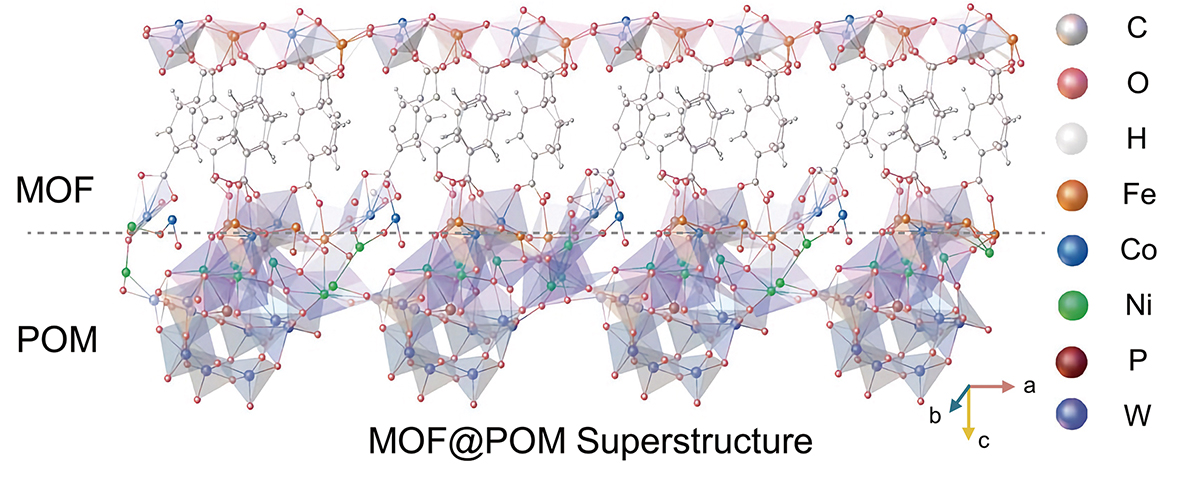
The MOF@POM superstructure enables water splitting with high activity and remarkable durability under industrial-level high current densities. (Graphic: SIC)

